| Revision as of 11:51, 23 September 2016 editBender the Bot (talk | contribs)Bots1,008,858 editsm →History: http→https for Google Books and Google News using AWB← Previous edit | Revision as of 16:49, 7 January 2017 edit undoInternetArchiveBot (talk | contribs)Bots, Pending changes reviewers5,388,258 edits Rescuing 1 sources and tagging 0 as dead. #IABot (v1.2.7.1)Next edit → | ||
| Line 65: | Line 65: | ||
| ==Dietary sources== | ==Dietary sources== | ||
| GLA is obtained from vegetable oils such as evening primrose ('']'') oil (EPO), ] seed oil, ], and ]. GLA is also found in varying amounts in edible ] seeds, oats, barley,<ref>{{cite journal | author = Qureshi AA | journal = Fed. Proc. | volume = 43 | pages = 2626 | year = 1984 |name-list-format=vanc | display-authors = etal}}</ref>{{full|date=November 2012}} and ]. Normal safflower ('']'') oil does not contain GLA, but a genetically modified GLA safflower oil available in commercial quantities since 2011 contains 40% GLA.<ref>{{cite journal|last=Nykiforuk|first=C.|author2=Shewmaker, C.|title=High level accumulation of gamma linolenic acid in transgenic safflower (Carthamus tinctorius) seeds|journal=Transgenic Research|date=19 August 2011|doi=10.1007/s11248-011-9543-5|volume=21|issue=2|pages=367–81|pmid=21853296}}</ref> Borage oil contains 20% GLA, evening primrose oil ranges from 8% to 10% GLA, and black-currant oil contains 15-20%.<ref>{{cite journal|last=Flider|first=Frank J|title=GLA: Uses and New Sources|journal=INFORM|date=May 2005|volume=16|issue=5|pages=279–282|url=http://www.adoniskish.com/PDF/Super%20Mix/gla.pdf}}</ref> | GLA is obtained from vegetable oils such as evening primrose ('']'') oil (EPO), ] seed oil, ], and ]. GLA is also found in varying amounts in edible ] seeds, oats, barley,<ref>{{cite journal | author = Qureshi AA | journal = Fed. Proc. | volume = 43 | pages = 2626 | year = 1984 |name-list-format=vanc | display-authors = etal}}</ref>{{full|date=November 2012}} and ]. Normal safflower ('']'') oil does not contain GLA, but a genetically modified GLA safflower oil available in commercial quantities since 2011 contains 40% GLA.<ref>{{cite journal|last=Nykiforuk|first=C.|author2=Shewmaker, C.|title=High level accumulation of gamma linolenic acid in transgenic safflower (Carthamus tinctorius) seeds|journal=Transgenic Research|date=19 August 2011|doi=10.1007/s11248-011-9543-5|volume=21|issue=2|pages=367–81|pmid=21853296}}</ref> Borage oil contains 20% GLA, evening primrose oil ranges from 8% to 10% GLA, and black-currant oil contains 15-20%.<ref>{{cite journal|last=Flider |first=Frank J |title=GLA: Uses and New Sources |journal=INFORM |date=May 2005 |volume=16 |issue=5 |pages=279–282 |url=http://www.adoniskish.com/PDF/Super%20Mix/gla.pdf |deadurl=yes |archiveurl=https://web.archive.org/web/20140112160125/http://www.adoniskish.com/PDF/Super%20Mix/gla.pdf |archivedate=2014-01-12 |df= }}</ref> | ||
| The human body produces GLA from ] (LA). This reaction is catalyzed by Δ<sup>6</sup>-] (D6D), an enzyme that allows the creation of a double bond on the sixth carbon counting from the carboxyl terminus. LA is consumed sufficiently in most diets, from such abundant sources as ]s and ]s. However, a lack of GLA can occur when there is a reduction of the efficiency of the D6D conversion (for instance, as people grow older or when there are specific dietary deficiencies) or in disease states wherein there is excessive consumption of GLA metabolites.<ref name="pmid8386433">{{cite journal |author=Horrobin DF (From the Efamol Research Institute. Kentville. Nova Scotia. Canada) |title=Fatty acid metabolism in health and disease: the role of delta-6-desaturase |journal=Am. J. Clin. Nutr. |volume=57 |issue=5 Suppl |pages=732S–736S; discussion 736S–737S |year=1993 |url=http://www.ajcn.org/cgi/reprint/57/5/732S.pdf|format=pdf|pmid=8386433 |doi=}}</ref>{{dubious|date=September 2013}} | The human body produces GLA from ] (LA). This reaction is catalyzed by Δ<sup>6</sup>-] (D6D), an enzyme that allows the creation of a double bond on the sixth carbon counting from the carboxyl terminus. LA is consumed sufficiently in most diets, from such abundant sources as ]s and ]s. However, a lack of GLA can occur when there is a reduction of the efficiency of the D6D conversion (for instance, as people grow older or when there are specific dietary deficiencies) or in disease states wherein there is excessive consumption of GLA metabolites.<ref name="pmid8386433">{{cite journal |author=Horrobin DF (From the Efamol Research Institute. Kentville. Nova Scotia. Canada) |title=Fatty acid metabolism in health and disease: the role of delta-6-desaturase |journal=Am. J. Clin. Nutr. |volume=57 |issue=5 Suppl |pages=732S–736S; discussion 736S–737S |year=1993 |url=http://www.ajcn.org/cgi/reprint/57/5/732S.pdf|format=pdf|pmid=8386433 |doi=}}</ref>{{dubious|date=September 2013}} | ||
Revision as of 16:49, 7 January 2017
This article is about gamma-Linolenic acid. For α-Linolenic acid, see alpha-Linolenic acid. For linoleic acid (no "n"), see linoleic acid.
| |||
| Names | |||
|---|---|---|---|
| IUPAC name all-cis-6,9,12-octadecatrienoic acid | |||
| Other names Gamma-linolenic acid, gamolenic acid, GLA | |||
| Identifiers | |||
| CAS Number | |||
| 3D model (JSmol) | |||
| ChEBI | |||
| ChEMBL | |||
| ChemSpider | |||
| ECHA InfoCard | 100.107.263 | ||
| IUPHAR/BPS | |||
| PubChem CID | |||
| UNII | |||
| CompTox Dashboard (EPA) | |||
InChI
| |||
SMILES
| |||
| Properties | |||
| Chemical formula | C18H30O2 | ||
| Molar mass | 278.436 g·mol | ||
| Appearance | Colorless oil | ||
| Pharmacology | |||
| ATC code | D11AX02 (WHO) | ||
| Except where otherwise noted, data are given for materials in their standard state (at 25 °C , 100 kPa).
| |||
Gamma-linolenic acid or GLA (γ-Linolenic acid), (INN and USAN gamolenic acid) is a fatty acid found primarily in vegetable oils. When acting on GLA, 5-lipoxygenase produces no leukotrienes and the conversion by the enzyme of arachidonic acid to leukotrienes is inhibited.
Chemistry
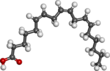
GLA is categorized as an n−6 (also called ω−6 or omega-6) fatty acid, meaning that the first double bond on the methyl end (designated with n or ω) is the sixth bond. In physiological literature, GLA is designated as 18:3 (n−6). GLA is a carboxylic acid with an 18-carbon chain and three cis double bonds. It is an isomer of α-linolenic acid, which is a polyunsaturated n−3 (omega-3) fatty acid, found in rapeseed canola oil, soy beans, walnuts, flax seed (linseed oil), perilla, chia, and hemp seed.
History
GLA was first isolated from the seed oil of evening primrose. This herbal plant was grown by Native Americans to treat swelling in the body. In the 17th century, it was introduced to Europe and became a popular folk remedy, earning the name king's cure-all. in 1919, Heiduschka and Lüft extracted the oil from evening primrose seeds and described an unusual linolenic acid, which they name γ-. Later, the exact chemical structure was characterized by Riley.
Although there are α- and γ- forms of linolenic acid, there is no β- form. One was once identified, but it turned out to be an artifact of the original analytical process.
Dietary sources
GLA is obtained from vegetable oils such as evening primrose (Oenothera biennis) oil (EPO), blackcurrant seed oil, borage seed oil, and hemp seed oil. GLA is also found in varying amounts in edible hemp seeds, oats, barley, and spirulina. Normal safflower (Carthamus tinctorius) oil does not contain GLA, but a genetically modified GLA safflower oil available in commercial quantities since 2011 contains 40% GLA. Borage oil contains 20% GLA, evening primrose oil ranges from 8% to 10% GLA, and black-currant oil contains 15-20%.
The human body produces GLA from linoleic acid (LA). This reaction is catalyzed by Δ-desaturase (D6D), an enzyme that allows the creation of a double bond on the sixth carbon counting from the carboxyl terminus. LA is consumed sufficiently in most diets, from such abundant sources as cooking oils and meats. However, a lack of GLA can occur when there is a reduction of the efficiency of the D6D conversion (for instance, as people grow older or when there are specific dietary deficiencies) or in disease states wherein there is excessive consumption of GLA metabolites.
Source of eicosanoids

From GLA, the body forms dihomo-γ-linolenic acid (DGLA). This is one of the body's three sources of eicosanoids (along with AA and EPA.) DGLA is the precursor of the prostaglandin PGH1, which in turn forms PGE1 and the thromboxane TXA1. Both PGE11 and TXA1 are anti-inflammatory; thromboxane TXA1, unlike its series-2 variant, induces vasodilation, and inhibits platelet consequently, TXA1 modulates (reduces) the pro-inflammatory properties of the thromboxane TXA2. PGE1 has a role in regulation of immune system function and is used as the medicine alprostadil.
Unlike AA and EPA, DGLA cannot yield leukotrienes. However, it can inhibit the formation of pro-inflammatory leukotrienes from AA.
Although GLA is an n−6 fatty acid, a type of acid that is, in general, pro-inflammatory, it has anti-inflammatory properties. (See discussion at Essential fatty acid interactions: The paradox of dietary GLA.)
Health and medicine
GLA has been promoted as medication for a variety of ailments including breast pain and eczema, in particular by David Horrobin (1939 – 2003), whose marketing of evening primrose oil was described by the British Medical Journal (BMJ) as ethically dubious – the substance was likely to be remembered as "a remedy for which there is no disease". In 2002 the UK's Medicines and Healthcare products Regulatory Agency withdrew marketing authorisations for evening primrose oil as an eczema remedy. Another single source suggests that Evening Primrose Oil with adjuvant vitamin E, may reduce breast pain.
Notes and references
- Yung-Sheng Huang, Vincent A. Ziboh (2001). Gamma-Linolenic Acid: Recent Advances in Biotechnology and Clinical Applications. AOCS Press. p. 259. ISBN 1-893997-17-0. Retrieved 2007-12-07.
- Eckey, EW (1954). Vegetable Fats and Oils (volume 123 of American Chemical Society monograph series). Reinhold. p. 542.
- Qureshi AA; et al. (1984). Fed. Proc. 43: 2626.
{{cite journal}}: Missing or empty|title=(help); Unknown parameter|name-list-format=ignored (|name-list-style=suggested) (help) - Nykiforuk, C.; Shewmaker, C. (19 August 2011). "High level accumulation of gamma linolenic acid in transgenic safflower (Carthamus tinctorius) seeds". Transgenic Research. 21 (2): 367–81. doi:10.1007/s11248-011-9543-5. PMID 21853296.
- Flider, Frank J (May 2005). "GLA: Uses and New Sources" (PDF). INFORM. 16 (5): 279–282. Archived from the original (PDF) on 2014-01-12.
{{cite journal}}: Unknown parameter|deadurl=ignored (|url-status=suggested) (help) - Horrobin DF (From the Efamol Research Institute. Kentville. Nova Scotia. Canada) (1993). "Fatty acid metabolism in health and disease: the role of delta-6-desaturase" (pdf). Am. J. Clin. Nutr. 57 (5 Suppl): 732S – 736S, discussion 736S–737S. PMID 8386433.
- King, Michael W. "Introduction to the Eicosanoids". The Medical Biochemistry Page. 1996–2013 themedicalbiochemistrypage.org, LLC. Retrieved 23 July 2013.
- Belch JJ, Hill A (2000). "Evening primrose oil and borage oil in rheumatologic conditions". Am. J. Clin. Nutr. 71 (1 Suppl): 352S – 6S. PMID 10617996. Retrieved 2007-12-07.
DGLA itself cannot be converted to LTs but can form a 15-hydroxyl derivative that blocks the transformation of arachidonic acid to LTs. Increasing DGLA intake may allow DGLA to act as a competitive inhibitor of 2-series PGs and 4-series LTs and, thus, suppress inflammation.
- Richmond, C. (2003). "David Horrobin". BMJ. 326 (7394): 885. doi:10.1136/bmj.326.7394.885.
- Williams, H. C (2003). "Evening primrose oil for atopic dermatitis: Time To Say Goodnight". BMJ. 327 (7428): 1358–9. doi:10.1136/bmj.327.7428.1358. JSTOR 25457999. PMC 292973. PMID 14670851.
- "Vitamin E and Evening Primrose Oil for Management of Cyclical Mastaglgia: A Randomized Pilot Study" (PDF). Alternative Medicine Review. Retrieved September 19, 2015.
{{cite journal}}: Cite journal requires|journal=(help)
| Leukotriene signaling modulators | |||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Receptor (ligands) |
| ||||||||||||||||
| Enzyme (inhibitors) |
| ||||||||||||||||
| Others | |||||||||||||||||