| Revision as of 21:28, 27 August 2009 edit24.37.41.212 (talk) →In other species← Previous edit | Revision as of 22:18, 27 August 2009 edit undoSaayiit (talk | contribs)477 edits Reverted 6 edits by 24.37.41.212; Overlinking. (TW)Next edit → | ||
| Line 42: | Line 42: | ||
| }} | }} | ||
| }} | }} | ||
| '''Urea''' or '''carbamide''' is an ] with the ] (]]<sub>2</sub>)<sub>2</sub>]]. The molecule has two ] ( |
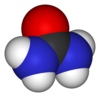
'''Urea''' or '''carbamide''' is an ] with the ] (]]<sub>2</sub>)<sub>2</sub>]]. The molecule has two ] (-NH<sub>2</sub>) residues joined by a ] (-CO-) ]. | ||
| Urea plays an important role in the ] of |
Urea plays an important role in the ] of nitrogen-containing compounds by animals, and is the main nitrogen-containing substance in the ] of ]s. Being solid, colourless, odorless, neither ] nor ], highly soluble in water, and relatively non-toxic, urea is widely used in ]s as a convenient source of nitrogen. Urea is also an important ] for the ]. The synthesis of this organic compound by ] 1828 from an inorganic precursor was an important milestone in the development of chemistry. | ||
| The terms '''urea''' and '''carbamide''' are also used for a class of ]s sharing the same ] |
The terms '''urea''' and '''carbamide''' are also used for a class of ]s sharing the same ] RR'N-CO-NRR', namely a carbonyl group flanked by two organic amine residues. Example include ], ], and ]. Ureas are closely related to ]s and related in structure to ]s, ]s, ]s, ]s, and ]s. | ||
| ==History== | ==History== | ||
| Urea was first discovered in |
Urea was first discovered in urine in 1773 by the ] chemist ]. | ||
| In |
In 1828, the ] chemist Friedrich Wöhler obtained urea by treating silver isocyanate with ammonium chloride in a failed attempt to prepare ammonium cyanate:<ref>{{cite book|title=Molecules That Changed The World|last=Nicolaou|first=Kyriacos Costa|authorlink=K. C. Nicolaou|coauthors=Tamsyn Montagnon|year=2008|publisher=Wiley-VCH|isbn=978-3-527-30983-2|pages=11}}</ref> | ||
| : |
: AgNCO + NH<sub>4</sub>Cl → (NH<sub>2</sub>)<sub>2</sub>CO + AgCl | ||
| This was the first time an |
This was the first time an organic compound was artificially synthesized from inorganic starting materials, without the involvement of living organisms. The results of this experiment implicitly discredited ]: the theory that the chemicals of living organisms are fundamentally different from inanimate matter. This insight was important for the development of ]. His discovery prompted Wöhler to write triumphantly to ]: "I must tell you that I can make urea without the use of kidneys, either man or dog. Ammonium cyanate is urea." For this discovery, Wöhler is considered by many the father of organic chemistry. | ||
| ==Physiology== | ==Physiology== | ||
| Urea is |
Urea is synthesized in the body of many organisms as part of the ], either from the oxidation of ] or from ]. In this cycle, ] groups donated by ammonia and L-] are converted to urea, while L-], ], L-], and L-] act as intermediates. Urea production occurs in the ] and is regulated by ]. Urea is found dissolved in blood (in the ] of 2.5 to 7.5 mmol/liter) and is excreted by the kidney as a component of urine. In addition, a small amount of urea is excreted (along with ] and water) in ]. | ||
| <table border=0> | <table border=0> | ||
| Line 71: | Line 71: | ||
| </table> | </table> | ||
| Aminoacids from ingested food which are not used for the synthesis of proteins and other biological substances are oxidized by the body, yielding urea and carbon dioxide, as an alternative source of energy.<ref>{{cite journal |author=Sakami W, Harrington H |title=Amino acid metabolism |journal= Annual Review of Biochemistry |volume=32 |issue= |pages=355–98 |year= 1963|pmid=14144484 |doi=10.1146/annurev.bi.32.070163.002035}}</ref> The oxidation pathway starts with the removal of the amino group by a ], the amino group is then fed into the urea cycle. | |||
| Ammonia (NH<sub>3</sub>) is another common byproduct of the metabolism of nitrogenous compounds. Ammonia molecules are smaller, more volatile and more mobile than urea's. If allowed to accumulate, ammonia would raise the pH in cells to toxic levels. Therefore many organisms convert ammonia to urea, even though this synthesis has a net energy cost. Being practically neutral (pK<sub>a</sub> close to zero) and highly soluble in water, urea is a safe vehicle for the body to transport and excrete excess nitrogen. | |||
| ===In humans=== | ===In humans=== | ||
| The ] is a vital part of |
The ] is a vital part of human metabolism. Besides its role as carrier of waste nitrogen, urea also plays a role in the ] of the ], that allows for reabsorption of water and critical ions from the excreted urine. Urea is reabsorbed in the ]s of the nephrons,<ref name=boron837> {{cite book |author=Walter F. Boron |title=Medical Physiology: A Cellular And Molecular Approaoch |publisher=Elsevier/Saunders |location= |year= |pages= |isbn=1-4160-2328-3 |oclc= |doi=}} Page 837 </ref> thus raising the ] in the medullary ] surrounding the ], which in turn causes water to be reabsorbed. By action of the ], some of this reabsorbed urea will eventually flow back into the thin ascending limb of the tubule, through the collecting ducts, and into the excreted urine. | ||
| This mechanism, which is controlled by the ], allows the body to create ] |
This mechanism, which is controlled by the ], allows the body to create ] urine, that has a higher concentration of dissolved substances than the ]. This mechanism is important to prevent the loss of water, to maintain ], and to maintain a suitable concentration of ] ions in the blood plasma. | ||
| ===In other species=== | ===In other species=== | ||
| In ] organisms the most common form of |
In ] organisms the most common form of nitrogen waste is ammonia, while land-dwelling organisms convert the toxic ammonia to either urea or ]. Urea is found in the urine of ]s and ]s, as well as some ]. ]s and ] ]s have a different form of nitrogen metabolism, that requires less ] and leads to nitrogen being excreted in the form of ]. It is noteworthy that ]s excrete ammonia but shift to urea production during ]. Despite the generalization above, the urea pathway has been documented not only in mammals and amphibians but in many other organisms as well, including birds, ]s, ]s, ]s, ], ], and even ]s. | ||
| ==Uses== | ==Uses== | ||
Revision as of 22:18, 27 August 2009
| |||
| Names | |||
|---|---|---|---|
| IUPAC name Diaminomethanal (as organic compound), Carbonyl diamide (as inorganic compound) | |||
| Other names Carbamide, carbonyl diamide, carbonyldiamine | |||
| Identifiers | |||
| CAS Number | |||
| 3D model (JSmol) | |||
| ChemSpider | |||
| ECHA InfoCard | 100.000.286 | ||
| E number | E927b (glazing agents, ...) | ||
| PubChem CID | |||
| RTECS number |
| ||
| CompTox Dashboard (EPA) | |||
SMILES
| |||
| Properties | |||
| Chemical formula | CH4N2O | ||
| Molar mass | 60.07 g/mol | ||
| Appearance | white odourless solid | ||
| Density | 1.32 g/cm | ||
| Melting point | 132.7–135 °C | ||
| Solubility in water | 108 g/100 ml (20 °C) 167 g/100 ml (40 °C) 251 g/100 ml (60 °C) 400 g/100 ml (80 °C) 733 g/100 ml (100 °C) | ||
| Acidity (pKa) | 0.18 | ||
| Basicity (pKb) | 13.82 | ||
| Structure | |||
| Dipole moment | 4.56 D | ||
| Hazards | |||
| Flash point | Non-flammable | ||
| Related compounds | |||
| Except where otherwise noted, data are given for materials in their standard state (at 25 °C , 100 kPa). Infobox references | |||
Urea or carbamide is an organic compound with the chemical formula (NH2)2CO. The molecule has two amine (-NH2) residues joined by a carbonyl (-CO-) functional group.
Urea plays an important role in the metabolism of nitrogen-containing compounds by animals, and is the main nitrogen-containing substance in the urine of mammals. Being solid, colourless, odorless, neither acidic nor basic, highly soluble in water, and relatively non-toxic, urea is widely used in fertilizers as a convenient source of nitrogen. Urea is also an important feedstock for the chemical industry. The synthesis of this organic compound by Friedrich Wöhler 1828 from an inorganic precursor was an important milestone in the development of chemistry.
The terms urea and carbamide are also used for a class of chemical compounds sharing the same functional group RR'N-CO-NRR', namely a carbonyl group flanked by two organic amine residues. Example include carbamide peroxide, allantoin, and hydantoin. Ureas are closely related to biurets and related in structure to amides, carbamates, diimides, carbodiimides, and thiocarbamides.
History
Urea was first discovered in urine in 1773 by the French chemist Hilaire Rouelle.
In 1828, the German chemist Friedrich Wöhler obtained urea by treating silver isocyanate with ammonium chloride in a failed attempt to prepare ammonium cyanate:
- AgNCO + NH4Cl → (NH2)2CO + AgCl
This was the first time an organic compound was artificially synthesized from inorganic starting materials, without the involvement of living organisms. The results of this experiment implicitly discredited vitalism: the theory that the chemicals of living organisms are fundamentally different from inanimate matter. This insight was important for the development of organic chemistry. His discovery prompted Wöhler to write triumphantly to Berzelius: "I must tell you that I can make urea without the use of kidneys, either man or dog. Ammonium cyanate is urea." For this discovery, Wöhler is considered by many the father of organic chemistry.
Physiology
Urea is synthesized in the body of many organisms as part of the urea cycle, either from the oxidation of amino acids or from ammonia. In this cycle, amino groups donated by ammonia and L-aspartate are converted to urea, while L-ornithine, citrulline, L-argininosuccinate, and L-arginine act as intermediates. Urea production occurs in the liver and is regulated by N-acetylglutamate. Urea is found dissolved in blood (in the reference range of 2.5 to 7.5 mmol/liter) and is excreted by the kidney as a component of urine. In addition, a small amount of urea is excreted (along with sodium chloride and water) in sweat.
|
|
|
|
Aminoacids from ingested food which are not used for the synthesis of proteins and other biological substances are oxidized by the body, yielding urea and carbon dioxide, as an alternative source of energy. The oxidation pathway starts with the removal of the amino group by a transaminase, the amino group is then fed into the urea cycle.
Ammonia (NH3) is another common byproduct of the metabolism of nitrogenous compounds. Ammonia molecules are smaller, more volatile and more mobile than urea's. If allowed to accumulate, ammonia would raise the pH in cells to toxic levels. Therefore many organisms convert ammonia to urea, even though this synthesis has a net energy cost. Being practically neutral (pKa close to zero) and highly soluble in water, urea is a safe vehicle for the body to transport and excrete excess nitrogen.
In humans
The handling of urea by the kidneys is a vital part of human metabolism. Besides its role as carrier of waste nitrogen, urea also plays a role in the countercurrent exchange system of the nephrons, that allows for reabsorption of water and critical ions from the excreted urine. Urea is reabsorbed in the inner medullary collecting ducts of the nephrons, thus raising the osmolarity in the medullary interstitium surrounding the thin ascending limb of the loop of Henle, which in turn causes water to be reabsorbed. By action of the urea transporter 2, some of this reabsorbed urea will eventually flow back into the thin ascending limb of the tubule, through the collecting ducts, and into the excreted urine.
This mechanism, which is controlled by the antidiuretic hormone, allows the body to create hyperosmotic urine, that has a higher concentration of dissolved substances than the blood plasma. This mechanism is important to prevent the loss of water, to maintain blood pressure, and to maintain a suitable concentration of sodium ions in the blood plasma.
In other species
In aquatic organisms the most common form of nitrogen waste is ammonia, while land-dwelling organisms convert the toxic ammonia to either urea or uric acid. Urea is found in the urine of mammals and amphibians, as well as some fish. Birds and saurian reptiles have a different form of nitrogen metabolism, that requires less water and leads to nitrogen being excreted in the form of uric acid. It is noteworthy that tadpoles excrete ammonia but shift to urea production during metamorphosis. Despite the generalization above, the urea pathway has been documented not only in mammals and amphibians but in many other organisms as well, including birds, invertebrates, insects, plants, yeast, fungi, and even microorganisms.
Uses
Agriculture
More than 90% of world production is destined for use as a nitrogen-release fertilizer. Urea has the highest nitrogen content of all solid nitrogenous fertilizers in common use (46.7%). Therefore, it has the lowest transportation costs per unit of nitrogen nutrient.
In the soil, it hydrolyses back to ammonia and carbon dioxide. The ammonia is oxidized by bacteria in the soil to nitrate which can be absorbed by the plants. Urea is also used in many multi-component solid fertilizer formulations. Urea is highly soluble in water and is, therefore, also very suitable for use in fertilizer solutions (in combination with ammonium nitrate: UAN), e.g., in 'foliar feed' fertilizers. For fertilizer use, granules are preferred over over prills because of their narrower particle size distribution which is an advantage for mechanical application.
The most common impurity of synthetic urea, biuret, must be present at less than 2%, as it impairs plant growth.
Urea is usually spread at rates of between 40 and 300 kg/ha, but actual spreading rates will vary according to farm type and region. It is better to make several small to medium applications at intervals to minimise leaching losses and increase efficient use of the N applied, compared with single heavy applications. During summer, urea should be spread just before, or during rain to reduce possible losses from volatilization (process wherein nitrogen is lost to the atmosphere as ammonia gas). Urea should not be mixed for any length of time with other fertilizers, as problems of physical quality may result.
Because of the high nitrogen concentration in urea, it is very important to achieve an even spread. The application equipment must be correctly calibrated and properly used. Drilling must not occur on contact with or close to seed, due to the risk of germination damage. Urea dissolves in water for application as a spray or through irrigation systems.
In grain and cotton crops, urea is often applied at the time of the last cultivation before planting. It should be applied into or be incorporated into the soil. In high rainfall areas and on sandy soils (where nitrogen can be lost through leaching) and where good in-season rainfall is expected, urea can be side- or top-dressed during the growing season. Top-dressing is also popular on pasture and forage crops. In cultivating sugarcane, urea is side-dressed after planting, and applied to each ratoon crop.
In irrigated crops, urea can be applied dry to the soil, or dissolved and applied through the irrigation water. Urea will dissolve in its own weight in water, but it becomes increasingly difficult to dissolve as the concentration increases. Dissolving urea in water is endothermic, causing the temperature of the solution to fall when urea dissolves.
As a practical guide, when preparing urea solutions for fertigation (injection into irrigation lines), dissolve no more than 30 kg urea per 100 L water.
In foliar sprays, urea concentrations of 0.5% – 2.0% are often used in horticultural crops. As urea sprays may damage crop foliage, specific advice should be sought before use. Low-biuret grades of urea should be used if urea sprays are to be applied regularly or to sensitive horticultural crops.
Like most nitrogen products, urea absorbs moisture from the atmosphere. Therefore it should be stored either in closed/sealed bags on pallets, or, if stored in bulk, under cover with a tarpaulin. As with most solid fertilizers, it should also be stored in a cool, dry, well-ventilated area.
Chemical industry
Urea is a raw material for the manufactuer of many important chemicals, such as
- Various plastics, especially the urea-formaldehyde resins.
- Various adhesives, such as urea-formaldehyde or the urea-melamine-formaldehyde used in marine plywood.
- Potassium cyanate, another industrial feedstock.
- Urea nitrate, an explosive.
Urea has the ability to trap many organic compounds in the form of clathrates. The organic compounds are held in channels formed by interpenetrating helices comprising of hydrogen-bonded urea molecules. This behaviour can be used to separate mixtures, and has been used in the production of aviation fuel and lubricating oils, and in the separation of paraffins.
As the helices are interconnected, all helices in a crystal must have the same molecular handedness. This is determined when the crystal is nucleated and can thus be forced by seeding. The resulting crystals have been used to separate racemic mixtures.
Automobile systems
Urea is used in SNCR and SCR reactions to reduce the NOx pollutants in exhaust gases from combustion, for example, from power plants and diesel engines. The BlueTec system, for example, injects water-based urea solution into the exhaust system. The ammonia produced by decomposition of the urea reacts with the nitrogen oxide emissions and is converted into nitrogen and water within the catalytic converter.
Other commercial uses
- A stabilizer in nitrocellulose explosives.
- A component of animal feed, providing a relatively cheap source of nitrogen to promote growth.
- A non-corroding alternative to rock salt for road de-icing, and the resurfacing of snowboarding halfpipes and terrain parks.
- A flavor-enhancing additive for cigarettes.
- A browning agent in factory-produced pretzels.
- An ingredient in some hair conditioners, facial cleansers, bath oils, skin softeners, and lotions.
- A reactant in some ready-to-use cold compresses for first-aid use, due to the endothermic reaction it creates when mixed with water.
- A cloud seeding agent, along with other salts.
- A flame-proofing agent, commonly used in dry chemical fire extinguisher charges such as the urea-potassium bicarbonate mixture.
- An ingredient in many tooth whitening products.
- An ingredient in dish soap.
- Along with ammonium phosphate, as a yeast nutrient, for fermentation of sugars into ethanol.
- An nutrient used by plankton in ocean nourishment experiments for geoengineering purposes.
- As an additive to extend the working temperature and open time of hide glue.
- As a solubility-enhancing and moisture-retaining additive to dye baths for textile dyeing or printing.
Laboratory uses
Urea in concentrations up to 10 M is a powerful protein denaturant as it disrupts the noncovalent bonds in the proteins. This property can be exploited to increase the solubility of some proteins.
A mixture of urea and choline chloride is used as a deep eutectic solvent, a type of ionic liquid.
Urea can serve as a hydrogen source, for subsequent power generation in a fuel cell. Urea present in urine/wastewater can be used directly (though bacteria normally quickly degrade urea.) Producing hydrogen by electrolysis of urea solution occurs at a lower voltage and uses less energy than by electrolysis of water.
Medical use
Urea is used in topical dermatological products to promote rehydration of the skin. If covered by an occlusive dressing, 40% urea preparations may also be used for nonsurgical debridement of nails. This drug is also used as an earwax removal aid.
Like saline, urea injection is used to perform abortions. It is also the main component of an alternative medicinal treatment referred to as urine therapy.
The blood urea nitrogen (BUN) test is a measure of the amount of nitrogen in the blood that comes from urea. It is used as a marker of renal function.
Urea labeled with carbon-14 or carbon-13 is used in the urea breath test, which is used to detect the presence of the bacteria Helicobacter pylori (H. pylori) in the stomach and duodenum of humans, associated with ulcers. The test detects the characteristic enzyme urease, produced by H. pylori, by a reaction that produces ammonia from urea. This increases the pH (reduces acidity) of the stomach environment around the bacteria. Similar bacteria species to H. pylori can be identified by the same test in animals such as apes, dogs, and cats (including big cats).
Hazards
Urea can be irritating to skin, eyes, and the respiratory tract. Repeated or prolonged contact with urea in fertilizer form on the skin may cause dermatitis.
Too high concentrations in the blood can cause damage to organs of the body. Ingestion of low concentrations of urea, such as are found in typical human urine, are not dangerous with additional water ingestion within a reasonable time-frame. Many animals (e.g., dogs) have a much more concentrated urine and it contains a higher urea amount than normal human urine; this can prove dangerous as a source of liquids for consumption in a life-threatening situation (such as in a desert).
Urea can cause algal blooms to produce toxins, and its presence in the runoff from fertilized land may play a role in the increase of toxic blooms.
The substance decomposes on heating above melting point, producing toxic gases, and reacts violently with strong oxidants, nitrites, inorganic chlorides, chlorites and perchlorates, causing fire and explosion hazard
Synthetic production
Urea is produced on a scale of some 100,000,000 tons per year worldwide.
Industrial methods
For use in industry, urea is produced from synthetic ammonia and carbon dioxide. Large quantities of carbon dioxide are produced during the manufacture of ammonia from coal or from hydrocarbons such as natural gas and petroleum-derived raw materials. This allows direct synthesis of urea from these raw materials.
The basic process, developed in 1922, is also called the Bosch-Meiser urea process after its discoverers. The various urea processes are characterized by the conditions under which urea formation takes place and the way in which unconverted reactants are further processed. The process consists of two main equilibrium reactions, with incomplete conversion of the reactants. The first is an exothermic reaction of liquid ammonia with dry ice to form ammonium carbamate (H2N-COONH4)::
- 2 NH3 + CO2 ↔ H2N-COONH4 ()
The second is an endothermic decomposition of ammonium carbamate into urea and water:
- H2N-COONH4 ↔ (NH2)2CO + H2O
Both reactions combined are exothermic.
Unconverted reactants can be used for the manufacture of other products, for example ammonium nitrate or sulfate, or they can be recycled for complete conversion to urea in a total-recycle process.
Urea can be produced as prills, granules, flakes, pellets, crystals, and solutions. Solid urea is marketed as prills or granules. The advantage of prills is that, in general, they can be produced more cheaply than granules. Properties such as impact strength, crushing strength, and free-flowing behaviour are, in particular, important in product handling, storage, and bulk transportation.
Laboratory processes
Ureas in the more general sense can be accessed in the laboratory by reaction of phosgene with primary or secondary amines, proceeding through an isocyanate intermediate. Non-symmetric ureas can be accessed by reaction of primary or secondary amines with an isocyanate.
Chemical properties
Molecular structure
The urea molecule is planar. Each carbonyl oxygen atom accepts four N-H-O hydrogen bonds. This dense and energetically favourable hydrogen-bond network is probably established at the cost of efficient molecular packing: The structure is quite open, the ribbons forming tunnels with square cross-section. The carbon in urea is described as sp2 hybridized, the C-N bonds have significant double bond character, and the carbonyl oxygen is basic compared to formaldehyde. Its high solubility is due to extensive hydrogen bonding with water: up to eight hydrogen bonds may form - two from the oxygen atom, one from each hydrogen atom and one from each nitrogen atom.
Reactions
Urea reacts with alcohols to form urethanes. Urea reacts with malonic esters to make barbituric acids.
References
- Nicolaou, Kyriacos Costa (2008). Molecules That Changed The World. Wiley-VCH. p. 11. ISBN 978-3-527-30983-2.
{{cite book}}: Unknown parameter|coauthors=ignored (|author=suggested) (help) - Sakami W, Harrington H (1963). "Amino acid metabolism". Annual Review of Biochemistry. 32: 355–98. doi:10.1146/annurev.bi.32.070163.002035. PMID 14144484.
- Walter F. Boron. Medical Physiology: A Cellular And Molecular Approaoch. Elsevier/Saunders. ISBN 1-4160-2328-3. Page 837
- Researchers develop urea fuel cell.
- newscientist.com - US set to track toxic algal blooms
- http://www.hillakomem.com/tag/ammonium-carbamate
External links
- MSDS sheet on urea
- Use of urea in hand dyeing
- use of urea as an automotive reagent to reduce harmful emissions (AdBlue)
| E numbers | |
|---|---|
|